
About Us Cibis Park
The third Cardiac Insufficiency Bisoprolol Study (CIBIS III) 13, 14 challenged commonly accepted practice by investigating whether a beta-blocker-first strategy was non-inferior to an ACE-inhibitor-first strategy in the initiation of CHF treatment in patients in NYHA classes II and III.
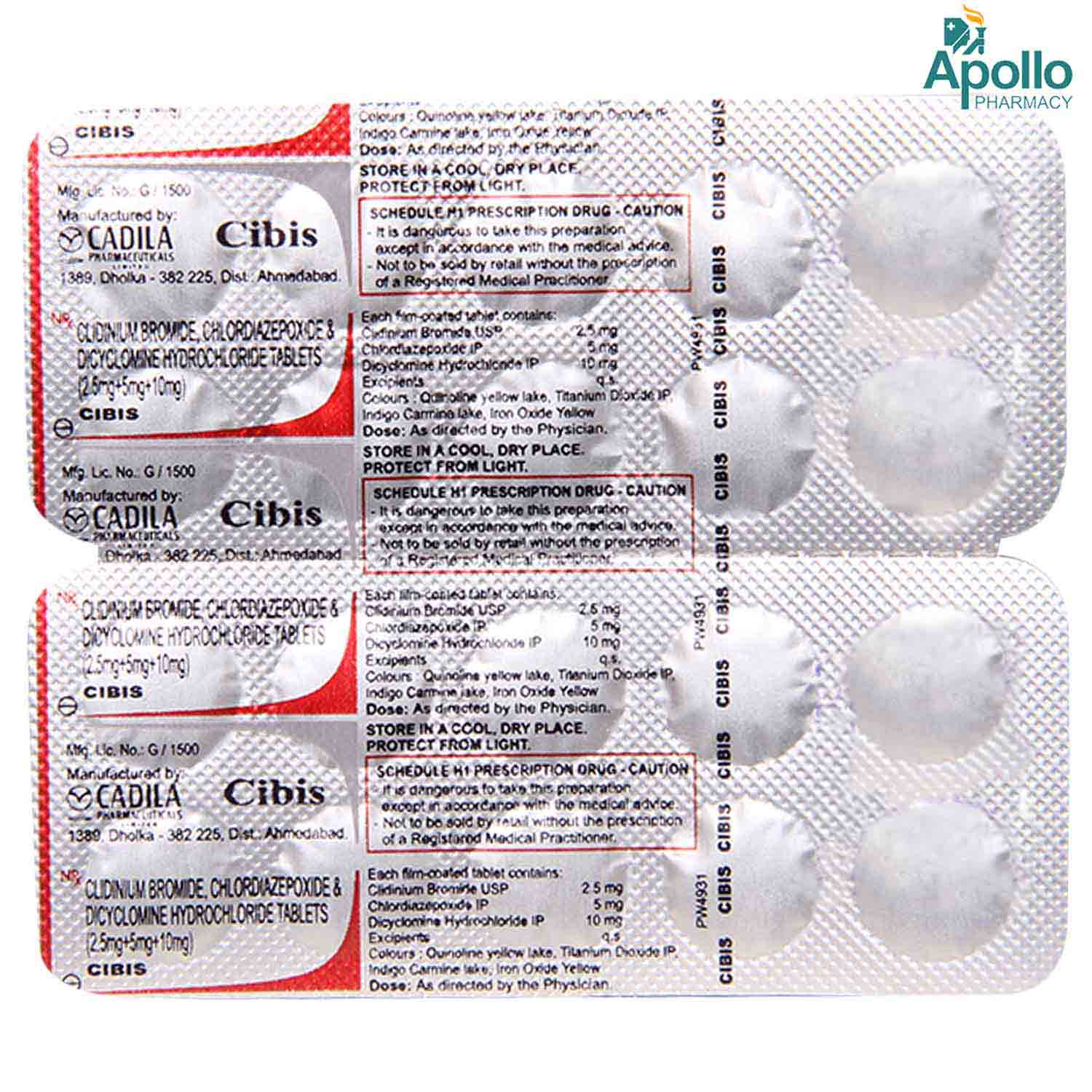
CIBIS TABLET Price, Uses, Side Effects, Composition Apollo Pharmacy
Findings: CIBIS-II was stopped early, after the second interim analysis, because bisoprolol showed a significant mortality benefit.. (156 [11.8%] vs 228 [17.3%] deaths with a hazard ratio of 0..

CIBIS Nine at CIBIS Park Cibis Park
Comparison of CIBIS meta-analysis with more recently published trials. The results of the CIBIS and CIBIS II meta-analysis were evaluated and combined with the results for total death of comparable older 5 and more recently published trials in which other β-blockers were tested 6, 7, 8 in a further meta-analysis (Figure 7).

CIBIS Tower Nine
In CIBIS, idiopathic dilated cardiomyopathy was diagnosed when no known cause of cardiomyopathy could be found. Patients were classified as having ischaemia when there was a typical history of coronary artery disease, a history of myocardial infarction, or the presence of a coronary stenosis greater than 70% shown by coronary angiography.

CIBIS Nine at CIBIS Park Construction Plus Asia
The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial Summary Background In patients with heart failure, β-blockade has improved morbidity and left-ventricular function, but the impact on survival is uncertain.

Cibis Tower 9
A recently derived 8-item version of the SIB—the SIB-8—which takes about 3 minutes to administer, may represent a more convenient tool for use in clinical practice. The current analyses further explored the SIB-8 scale with respect to its validity and sensitivity. Methods

Nine Cibis Park
Findings CIBIS-II was stopped early, after the second interim analysis, because bisoprolol showed a significant mortality benefit. All-cause mortality was significantly lower with bisoprolol than on placebo (156 [11·8%] vs228 [17·3%] deaths with a hazard ratio of 0·66 (95% CI 0·54-0·81, p<0·0001). There were significantly fewer

About Us Cibis Park
CIBIS-II Investigators and Committees. The Cardiac Insufficiency Bisoprolol Study II (CIBIS II): a randomised trial.. 8. Sackner-Bernstein J, Krum H, Goldsmith RL, et al. Should worsening heart.

Eight Cibis Park
The landmark Cardiac Insufficiency Bisoprolol Study (CIBIS) II was the first trial to show a mortality benefit in moderate-to-severe stable CHF (NYHA III or IV; LVEF ≤35%). 8 It was followed by the Metoprolol CR/XL Randomized Intervention Trial in Congestive Heart Failure 9 (MERIT-HF) in mild-to-moderate, stable, systolic CHF, and by the.

Broadway Malyan CIBIS
SPECIFICATION AT A GLANCE 1.1 Ha land area ± 17.806 sqm semigross ± 3,751 sqm typical semigross 7 typical floor 250 kg/sqm floor loading GF - 1st Floor 3,751 sqm of semigross 3,362 sqm of nett area 2nd Floor 2,341 sqm of semigross 2,341 sqm of nett area 3rd Floor 3,751 sqm of semigross 3,125 sqm of nett area 5th Floor 3,751 sqm of semigross 3,390

CIBIS Nine at CIBIS Park Cibis Park
Bisoprolol was the first beta-blocker shown to have beneficial effects on outcomes in the Cardiac Insufficiency Bisoprolol Study II (CIBIS-II) ( CIBIS-II Investigators and Committees 1999 ). The aim of this article is to review its main pharmacological characteristics with respect to its use in the patients with HF. Go to:

CIBIS Nine, Jakarta
Bisoprolol was evaluated in the CIBIS-II trial, leading to all-cause mortality of 8.8% versus 13.2% in the placebo group (P < 0.0001).. (P < 0.001) and 2.8% (P < 0.001), respectively. 5 The total daily strength of the combination product used in the trial offered bioavailability similar to 320 mg valsartan. Although this is the desired.

Gallery of Cilandak Bisnis Square (CIBIS) Masterplanning Project
1. CIBIS-II Investigators and Committees The Cardiac Insufficiency Bisoprolol II (CIBIS-II): a randomised trial. Lancet. 1999; 353: 9-13 View in Article Scopus (4219) PubMed Summary

RetailLink Cibis Park
The investigators randomized 1010 patients with mild to moderate heart failure, and an ejection fraction <35%, to open-label monotherapy with either bisoprolol (target dose 10 mg/day) or enalapril (target dose 10 mg twice daily) for 6 months followed by uptitration of the second drug for 6 to 24 months.

Cibis Park, Tempat Rekreasi Gratis Warga Jakarta Rumika's Journey
Bisoprolol fumarate is a highly selective beta-1 receptor blocker. Bisoprolol has been extensively studied in three large mortality trials in stable chronic heart failure (CHF) patients. The CIBIS trial enrolled 641 patients and demonstrated the good tolerability of bisoprolol in a large CHF population, without evidence for any harmful effect.

CIBIS Nine at CIBIS Park Cibis Park
The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial 1999 Jan 2;353 (9146):9-13. PMID: 10023943 In patients with heart failure, beta-blockade has improved morbidity and left-ventricular function, but the impact on survival is uncertain.